Blog Post #14 – In this post to The Confined Space Training Blog, we will examine another reason a confined space identified at your workplace should be classified as a permit-required confined space. And that is when a confined space contains a liquid or material that could potentially engulf the entrant.
What is Engulfment in a Confined Space?
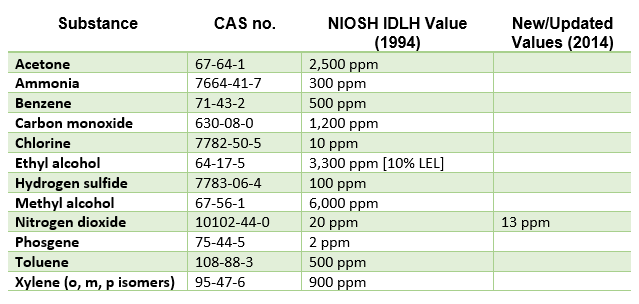
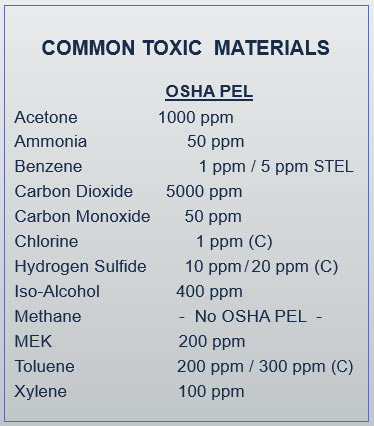
The Federal OSHA definition of engulfment can be found in 1910.146 paragraph B, and includes confined spaces where the entrant could suddenly be surrounded or captured by liquid, trapped inside the space, and drown in the liquid.

Engulfment by liquid could occur in sanitary and storm sewers, as well as in any pipelines, utility vaults, or process tanks that could suddenly be filled with liquid.
One other form of engulfment is possible whenever a confined space contains a finely divided solid material, such as pellets, chips, grains, granules, dusts, or powders, that can be aspirated by the entrant once engulfed. his could cause death by filling or plugging the respiratory system. In other cases, the material surrounding the trapped entrant can exert enough force on the body to cause death by strangulation, constriction, or crushing. Regardless of the mode of engulfment, these are all a gruesome way to die!
Confined spaces that contain potential engulfment hazards include, but are not limited to, grain elevators and storage bins, as well as other confined spaces that contain materials such as flour, sawdust, powdered cement, or finely pelletized materials.
How Does Engulfment in a Solid Material Occur Inside a Confined Space?
The illustration below demonstrates how a finely divided material can appear from the top to fill a confined space when, in actuality, a hollow void area could develop beneath the surface. This condition is often referred to as “bridging.”
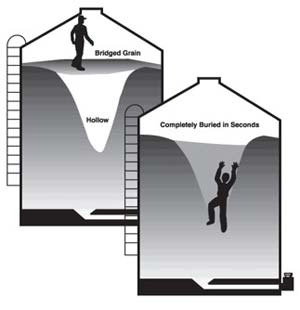
The weight of the employee can cause the bridged material to collapse, and the worker can fall into the void area and be covered by the flowing material.
In this second illustration, the employee is engulfed when the material, which has clung to the side of the wall inside the confined space, collapses and covers the employee.

Recap of Engulfment in a Confined Space
So in review, any confined space containing, or with the reasonable potential to be quickly filled with, a liquid or finely divided material that can engulf the entrant must be treated as a permit-required confined space.
Please provide your feedback and/or questions about this blog post on engulfment hazards in confined spaces in the comments section below. Then be sure to come back later and read our next blog post (#15), where we will discuss additional hazards that can trigger the permit-requirements in confined spaces.
And as always, I ask that you take a moment to spread the word about our confined space training blog by sharing a link to this post with others in your network so they can benefit from this information. Thanks – Curtis